Independent Biopharma Stock Research
Better intelligence tools for regulatory, financial, and clinical trial catalysts
Discover Trading Opportunities at Every Stage of the Drug Development Pipeline
| Product | Preclinical | Phase 1 | Phase 2 | Phase 3 | FDA Review | Marketed |
|---|---|---|---|---|---|---|
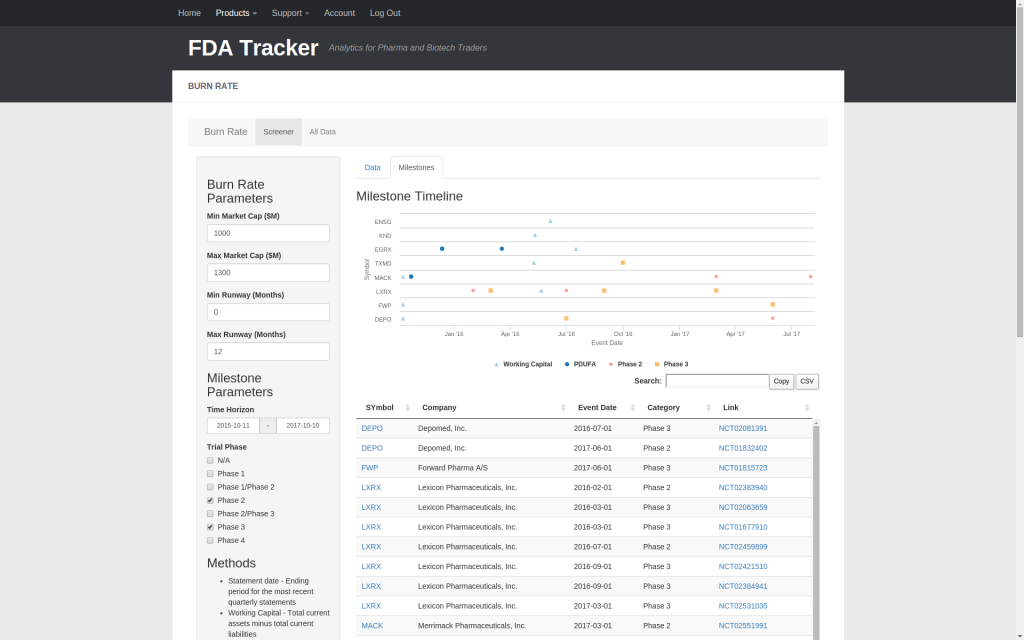
| Burn Rate | ✅ | ✅ | ✅ | ✅ | ✅ | ✅ |
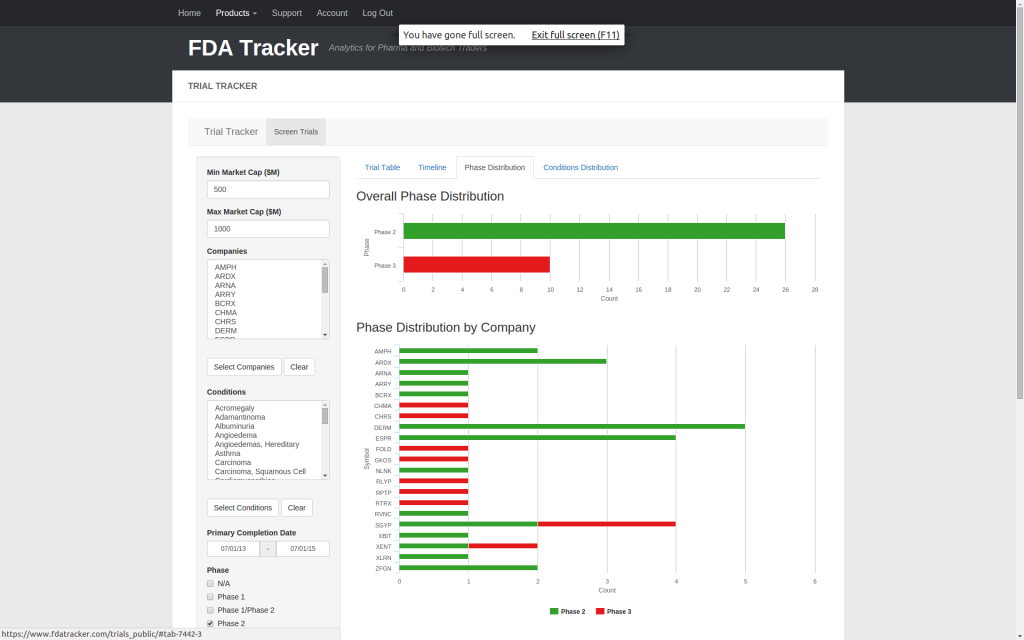
| Trial Tracker | ✅ | ✅ | ✅ | |||
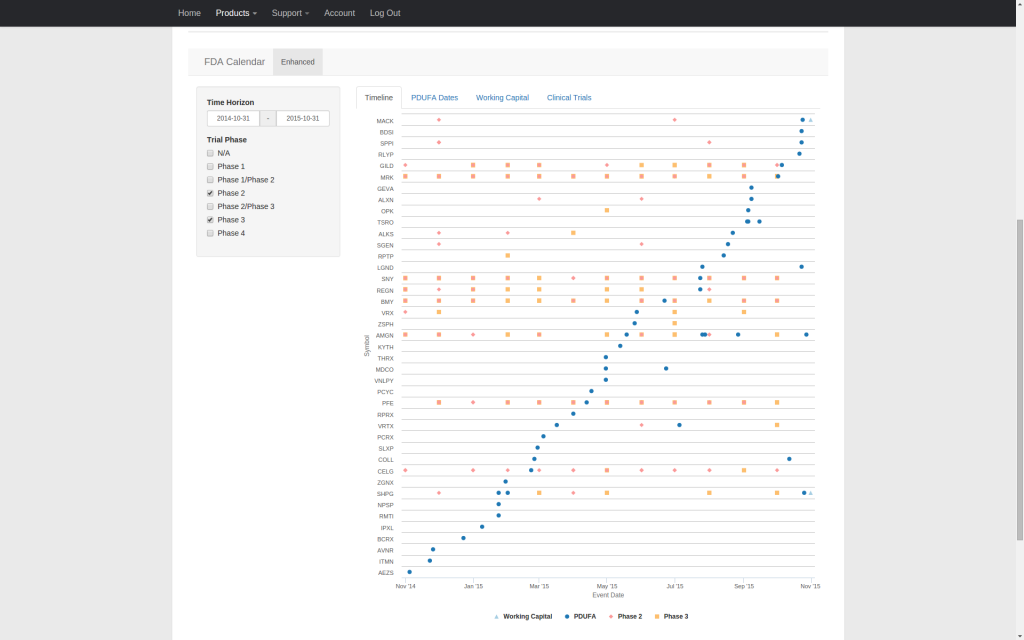
| FDA Calendar | ✅ | |||||
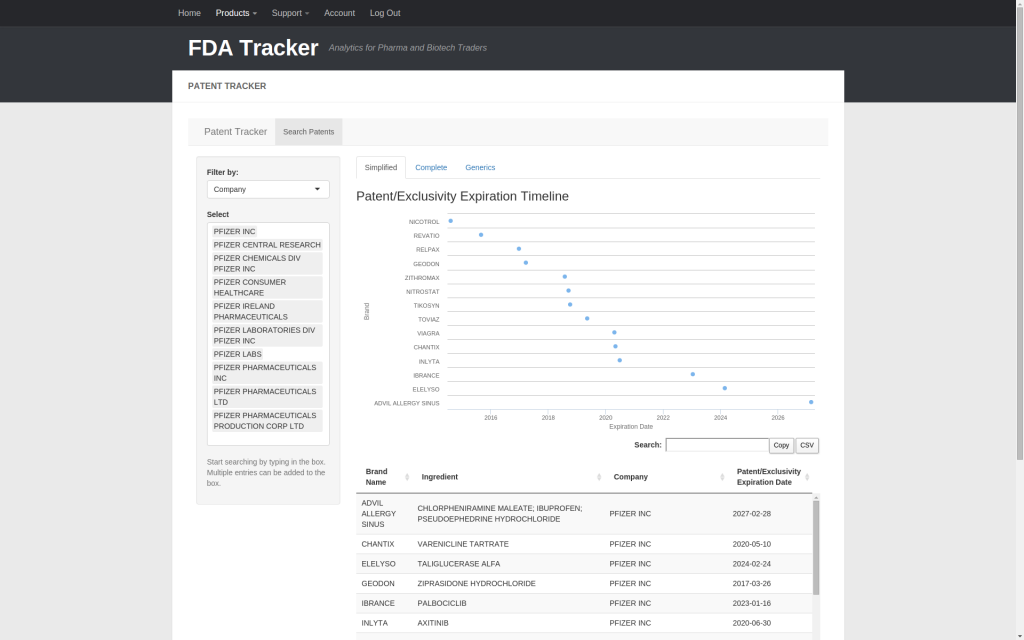
| Patent Tracker | ✅ |